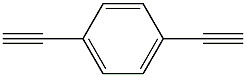
C10H6 1 4 Diethynylbenzene CAS 935-14-8 Yellow Powder 126.15 MW 98% Purity
-
High Light
methenolone enanthate powder
,testosterone propionate powder
-
CAS935-14-8
-
Product Name1,4-Diethynylbenzene
-
MFC10H6
-
MW126.15
-
AppearanceYellow Powder
-
Purity98%
-
Place of OriginChina
-
Brand NameRUN
-
CertificationISO9001
-
Model NumberRUN-AH
-
Minimum Order Quantity10g
-
Priceinquiry
-
Packaging Details10g 50g 100g 500g 1kg
-
Delivery Time3-7 days
-
Payment TermsT/T, , MoneyGram, BTCcoin
-
Supply Ability1kg --100kg
C10H6 1 4 Diethynylbenzene CAS 935-14-8 Yellow Powder 126.15 MW 98% Purity
C10H6 1 4 Diethynylbenzene CAS 935-14-8 Yellow Powder 126.15 MW 98% Purity
Specification
The 1,4-Diethynylbenzene, with the CAS registry number 935-14-8, is also known as Benzene, 1,4-diethynyl-. It belongs to the product categories of Miscellaneous Natural Products; Acetylenes; Acetylenic Hydrocarbons Having Benzene Ring; Building Blocks for Liquid Crystals; Chalcones, etc. (Building Blocks for Liquid Crystals); Fluorenes, etc. (reagent for high-performance polymer research); Functional Materials; Reagent for High-Performance Polymer Research; Alkynes; Organic Building Blocks; Terminal. This chemical's molecular formula is C10H6 and molecular weight is 126.15. What's more, its IUPAC name is the same with its product name. It should be stored in a cool, dry and well-ventilated place.
Physical properties about 1,4-Diethynylbenzene are: (1)ACD/LogP: 3.23; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 3.23; (4)ACD/LogD (pH 7.4): 3.23; (5)ACD/BCF (pH 5.5): 167.71; (6)ACD/BCF (pH 7.4): 167.71; (7)ACD/KOC (pH 5.5): 1361.27; (8)ACD/KOC (pH 7.4): 1361.27; (9)#H bond acceptors: 0; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 0; (12)Polar Surface Area: 0 Å2; (13)Index of Refraction: 1.567; (14)Molar Refractivity: 40.875 cm3; (15)Molar Volume: 125.075 cm3; (16)Surface Tension: 43.35 dyne/cm; (17)Density: 1.009 g/cm3; (18)Flash Point: 51.851 °C; (19)Enthalpy of Vaporization: 40.188 kJ/mol; (20)Boiling Point: 182.812 °C at 760 mmHg; (21)Vapour Pressure: 1.085 mmHg at 25 °C.
Preparation of 1,4-Diethynylbenzene: this chemical can be prepared by 1,4-Bis-(trimethylsilyl-aethinyl)-benzol. This reaction needs reagent K2CO3 and solvent methanol at temperature of 20 °C. The yield is 97 %.
Uses of 1,4-Diethynylbenzene: it is used to produce other chemicals. For example, it can react with cis-1,2-dichloro-ethene to get 1,4-Bis-(4-chloro-but-3-en-1-ynyl)-benzene. The reaction occurs with reagents n-BuNH2, copper(II) acetate, Pd(PPh3)2Cl2 and solvent toluene at ambient temperature. The yield is 70 %.
When you are dealing with this chemical, you should be very careful. This chemical may cause inflammation to the skin or other mucous membranes. It may catch fire in contact with air, only need brief contact with an ignition source and have a very low flash point or evolve highly flammable gases in contact with water. It may cause sensitisation by skin contacting. In addition, the production is harmful to aquatic organisms and may cause long-term adverse effects in the aquatic environment. Therefore, you should wear suitable gloves and eye/face protection.
You can still convert the following datas into molecular structure:
(1) SMILES: C#Cc1ccc(cc1)C#C
(2) InChI: InChI=1S/C10H6/c1-3-9-5-7-10(4-2)8-6-9/h1-2,5-8H
(3) InChIKey: MVLGANVFCMOJHR-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | unreported | 4500mg/kg (4500mg/kg) | BEHAVIORAL: ALTERED SLEEP TIME (INCLUDING CHANGE IN RIGHTING REFLEX) LUNGS, THORAX, OR RESPIRATION: RESPIRATORY STIMULATION |
Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 30(5), Pg. 59, 1986. |